Introduction
The aim of this article is to describe and discuss some differences between the short peripheral intravenous catheter (SPIVC) and the long peripheral intravenous catheter (LPIVC) inserted using the Seldinger Technique. It will consider how these differences might impact on dwell time, complications, and the patient experience. In this article, the definitions defined by AVA will be used.
- ‘Short peripheral intravenous catheter (short PIVC): an over-the-needle catheter with a hollow metal stylet (needle) positioned inside the catheter, generally inserted in superficial veins’
- ‘Long peripheral intravenous catheter (long PIVC): inserted in either superficial or deep peripheral veins and offers an option when a short PIVC is not long enough to adequately cannulate the available vein. A long PIVC can be inserted via traditional over-the-needle technique or with more advanced procedures, such as Seldinger and accelerated Seldinger techniques’
Additionally, the terms extended dwell catheter (EDC) and midline catheter (MC) will be used interchangeably for a long peripheral intravenous catheter (LPIVC).
Background
The insertion of devices into the venous system to either take blood or to deliver therapy has been performed since the venous system was discovered in the fifteenth century (Rivera et al., 2005). Subsequently, techniques, technologies, and methods of intravenous (IV) access and therapy have advanced rapidly. The evolution and development of IV therapy into the twentieth century has led to the evolution of vascular access devices (VAD) necessary to deliver these therapies. This article will focus on peripheral VAD.
Short Peripheral Intravenous Catheters: often called cannulas, venflons, IVs or drips and are used in most clinical settings. They are required for many reasons including:
- Delivery of peripherally IV therapies
- Blood or blood products
- Isotonic fluids and medications
- Hydration
- Administration of contrast media for diagnostic test
- Delivery of chemotherapy
Research has shown that SPIVCs are often used as a default device, despite not always being the most appropriate device for some patients (Hallam et al, 2016). It is also recognised that SPIVCs are not without the risk of failure. A very recent study of 5325 patients describes a failure rate of 54.05% (2889 / 5345). The most common complications noted in this study were phlebitis (16.3% and infiltration / extravasation (13.8%) (Chen, 2022). SPIVCs often fail or dislodge between days three and five. Therefore they are not recommended for peripheral IV therapies that are predicted to be necessary for periods longer than this (Chopra et al., 2015; Helm et al., 2015; Denton, 2016; Gorski, 2016).
To bridge the gap between PIVCs and central venous catheters such as peripherally inserted central caterers (PICC), LPIVCs were introduced into practice in the 1980s. Subsequently, the use of LPIVC has continued to grow in popularity as an alternative to the SPIVC.
LPIVC are used for the administration of blood, fluid, and medication when the therapy is expected to last between one and four weeks. They can be used where patients present with poor peripheral venous access or when the use of a central venous catheter is contraindicated. According to Gorski, (2016) the LPIVC provides venous accessibility along with an easy, less hazardous insertion.
There are some key differences between these two devices. These will now be discussed.
Insertion techniques
One of the main differences between the devices is the method of insertion.
Over the needle method
Cannulas are inserted using an over the needle method detailed below:
Skill acquisition
The cannulation of a vein can be very challenging, especially if the veins are small or tortuous. According to Marshburn. Engelke, and Swanson (2009), SPIVC insertion is considered the most difficult skill for nurses to master. Concern has been expressed about the potential risk to patients because of a lack of competence in the skill (Zamanzadeh et al. 2015; Milutinovic, Simin, and Zec, 2015 and Meeder et al. 2016). According to Coomarasamy, (2014) further investment in training and certification of nurses to undertake cannulation might result in improvements.
As well as nurses, doctors are often responsible for the insertion of SPIVCs. A study by Carr et al. (2011) suggested that interns (junior doctors) are poorly prepared for this procedure. Surprisingly, the interns in this study showed limited understanding of whether SPIVC insertion was a clean or aseptic technique and had little knowledge of the potential side effects of peripheral cannulation. Subsequently, the success rate for first time canulation is low and the complication rate of peripheral cannulas is unacceptably high (Helm, 2015, van Loom, 2018). This was also highlighted in a study by Mörgeli, et al (2022) which suggested that up to 25% of adult patients and 50% of paediatric patients experience a first – attempt cannulation failure rate.
Seldinger technique
The Seldinger technique (ST) is often used for the insertion of LPIVC. The ST provides a safer alternative to the traditional surgical cut–down technique and was first described by Sven-Ivar Seldinger (Seldinger, 1953). The technique is simple, requiring only a thin-walled introducer needle, guide wire and a catheter. The technique involves the target vein being punctured with a sharp hollow needle; following this, a guide wire is passed through the needle and into the vessel. Leaving the guidewire within the vessel, the needle is withdrawn. Finally, the catheter fed over the wire directly into the vessel.
Skill acquisition
Because LPIVC / midline catheters are ‘new’ devices, there seems to continue to be a more structured approach to education and training.
There are few studies that look at the ease and success of the Seldinger technique compared to others insertion techniques. However, an early study by Beards et al. (1994) claimed that the use of the direct puncture was associated with a significantly higher failure rate (23%) than the use of a catheter with a separate wire, namely the Seldinger Technique. Although this study focused on arterial catheters the findings are transferable to all vascular access.
Methods to improve venous access success
Cannulation of veins for both SPIVC and LPIVC is not always easy. Therefore, everything should be done to help achieve first time success.
The procedure for SPIVC insertion is typically taught and performed using palpation and unassisted visualisation. These methods give little information about vein quality, size, depth, and health. More recently, clinicians are beginning to use vein visualisation technology and/or ultrasound guidance to improve the success rate of peripheral venous cannulation.
Vein visualisation technology
Chiao et al. (2013) evaluated an infrared vein finding technology device to identify cannulation sites. They compared success using the device with success using unassisted eye site. They concluded that African American or Asian ethnicity and obesity were associated with decreased vein visibility. However, the visibility of veins suitable for cannulation increased for all subgroups using an infrared device.
Firooz et al. (2022) published a meta-analysis which aimed to assess the effect of transluminal devices on first attempt success rate, mean number of attempts and mean duration of successful PIVC placement in children. Based on the findings from four randomised controlled trials (RCT), the use of transilluminator devices significantly increased the first-time success rate and peripheral venous catheter placement. In addition, the results of two non RCTs showed transilluminator devices insignificantly decreased the first attempt success rate of peripheral venous catheter placement.
Ultrasound Guided Venous Access
Ultrasound guided cannulation has been shown to increase first time success rates, reduce the number of attempts to successful cannulation, reduce the time to get adequate access and increase patient satisfaction (van-Loon, 2018). Therefore, to increase the success of peripheral cannulation and reduce patient pain and dissatisfaction, technology should be considered.
Insertion Sites
The site of insertion often varies between these two devices, and this can influence device dwell time.
Back of the hand
The dorsal arch at the back of the hand has been a traditional area for SPIVC placement for many years. Clinicians are often taught to place them in this area and continue to do so. Subsequently, this has become custom and practice. What we now know is that the back of the hand is an area were there is a lot of movement. It is an area of flexion which can lead to mechanical phlebitis and catheter failure. The antecubital fossa is another area where SPIVC are often inserted. This is because it is an area where a large vein is often palpable. However, this site is also an area of flexion and is an uncomfortable and restrictive for patients. It is well documented that areas of flexion should be avoided for vascular access device insertion (Rickard, Webster, Wallice, 2012; Marsh, Webster, Larsen, et al., 2018a).
Forearm upper arm
Forearm placement of both SPIVC and LPIVC is a good alternative to using the dorsal arch of the back of the hand. If ultrasound guidance is not available, veins must be visible and palpable, straight, and bouncy. It must be assured that the catheter does not cross any area of flexion or terminate at the antecubital fossa.
Mid upper arm
Research by Dawson (2015) demonstrated that placement of devices in the middle of the upper arm reduced the risk of mechanical phlebitis and infection. This area is therefore regarded as optimal for midlines inserted in the upper arm.
Integrated extension set
The majority of SPIVC do not have an integrated extension set attached. In some cases, the needle free device (NFD) is attached directly to the end of the peripheral cannula. A degree of force is required to depress the inner membrane of the NFD, and this causes movement within the vein intima. This friction can lead to mechanical phlebitis (inflammation of the inner layer of the vein). Subsequently, this can result in device failure. To reduce the risk of this complication, it is recommended that an extension is attached to the end of SPIVC (Dougherty and Lamb, 2008; Hadaway, 2011).
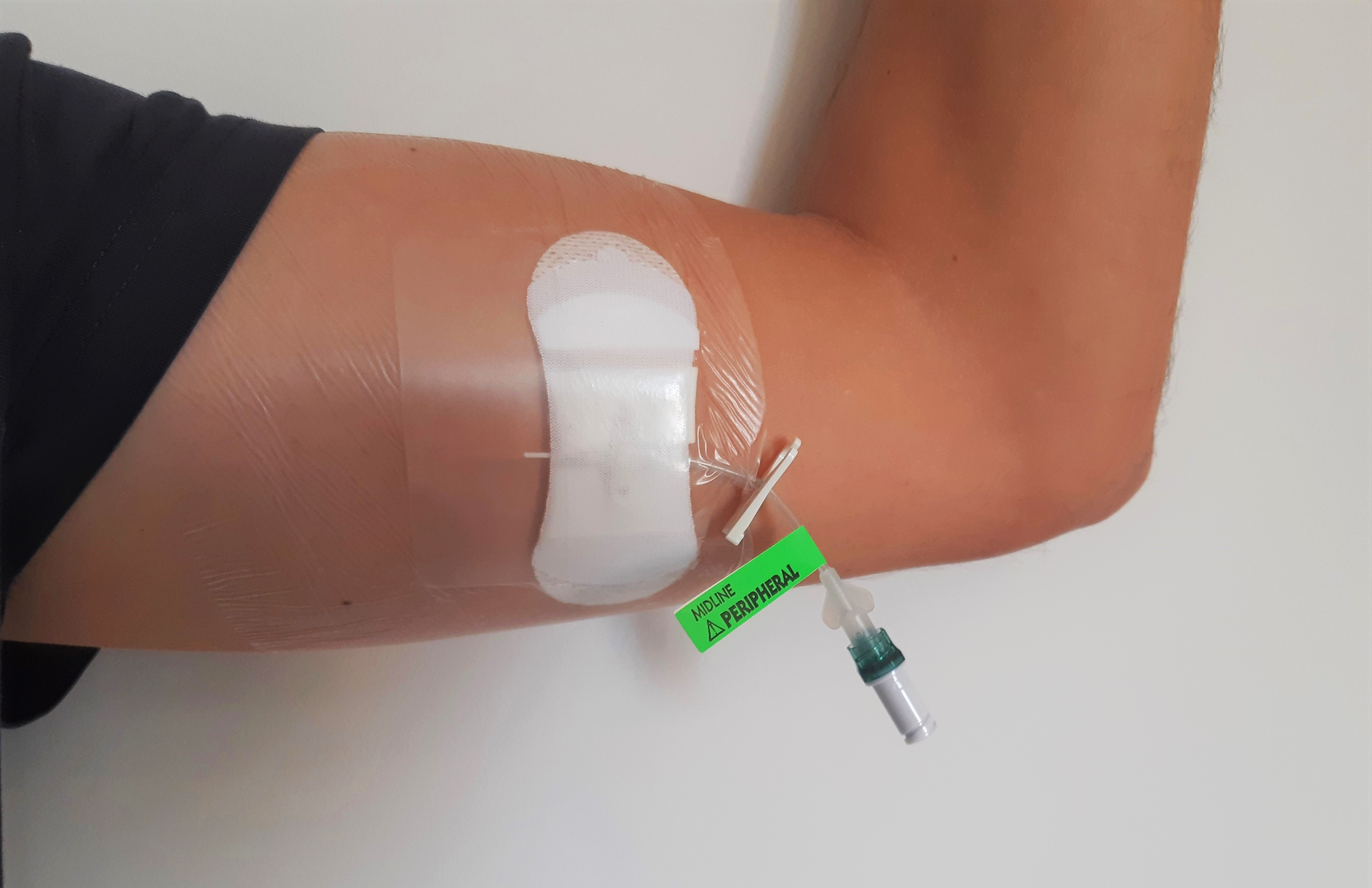
Some LPIVC have integrated extension sets which means that this risk is averted.
The use of engineered stabilisation devices is also possible due to the wings attached to many of the LPIVCs.
Device length
A short peripheral intravenous catheter length is described as less than 3 inches / 7.62 cm long. The length of a LPIVC is between 7.5 to 25 cms. Because of the length of LPIVCs, rather than being placed in the hand, they are either inserted into veins in the forearm (basilic or cephalic veins) or the upper arm (basilic, brachial or cephalic). Therefore, LPIVC allow a better vein purchase. To reduce the risk of dislodgement, it is important to ensure that length will reside in at least two thirds of the catheter length resides within the vein (Bahl et al., 2019). This reduces the risk of catheter dislodgement. Therefore, a longer device might be necessary when inserting them into the deeper veins of the upper arm. The veins of obese patients may also be deeper so the device length would have to accommodate this. Using a longer device also allows placement in the upper deeper arm veins which also increases the catheter to vein ratio, resulting in a reduced risk of thrombosis (Bahl, 2019).
Catheter material
Vascular access device tip termination is important if damage to the vein intima is to be avoided. Rubbing and subsequent trauma to the vein intima can result in mechanical phlebitis. Due to improvements in catheter materials this risk has been reduced. Most vascular access devices are made from either silicone or polyurethane (PUR). Some of the LPIVC have a high ratio of soft polymers. This means that once in contact with blood there is a softening effect of the PUR polymer. This helps to reduce vein trauma, increase tolerance, and lowers the incidences of mechanical phlebitis and thrombosis.
The site of insertion, catheter material, catheter design and length of the LPIVC appear to have an influence on catheter dwell time.
Dwell time
Literature suggests that SPIVC have a dwell time of approximately 2.3 to 4.2 days (DeVries et al., 2016; Larsen et al. 2021), whereas a LPIVC has been claimed to dwell for 7.7 to 16.4 days (Fabiani et al., Moureau et al., 2015).
Ported catheters
SPIVC can be ported or non-ported. In the United Kingdom, ported SPIVCs are still used widely. These have been associated with increase dislodgement rates and increased infection. Eastterson, Hoddinott Harrison (2011) introduced non-ported SPIVC to address these complications. Eight months following the introduction of non – ported safety SPIVC there was a decrease of 53% and 35% reports in the number of methicillin-resistant Staphylococcus aureus and healthcare-acquired infection cases, respectively, compared with the same period prior to implementation. Audits results also demonstrated considerable improvements in practices relating to cannula care following implementation of the new SPIVC. It appears that globally the use of ported SPIVCs might continue to decrease into the future.
Conclusion
SPIVC and LPIVC both allow the administration of peripherally compatible intravenous therapy. SPIVC are inserted frequently and have become a ‘go to’ device for many. It is now recognised that SPIVC have an endemically high rate of complications. Newer LPIVC are being used more frequently globally. Early research suggests that these devices are good alternatives to SPIVC due to improved dwell times (Adam et al. 2016). Finally, it is suggested the Midline catheters appear to be superior for patients with difficult vascular access or receiving prolonged intravenous therapy (Marsh et al. 2022).
This paper has discussed some of the main differences between the two devices and suggested why these differences might influence dwell time.
References
Adams DZ, Little A, Vinsant C, Khandelwal S. The Midline Catheter: A Clinical Review. J Emerg Med. 2016 Sep;51(3):252-8. doi: 10.1016/j.jemermed.2016.05.029. Epub 2016 Jul 5. PMID: 27397766.
Bahl, A., Karabon, P., & Chu, D. (2019). Comparison of Venous Thrombosis Complications in Midlines Versus Peripherally Inserted Central Catheters: Are Midlines the Safer Option?. Clinical and applied thrombosis/hemostasis : official journal of the International Academy of Clinical and Applied Thrombosis/Hemostasis, 25, 1076029619839150. https://doi.org/10.1177/1076029619839150
Baltalalarli A, Adiguzel E, Gurses E, Coskun E, Subclavian vein cannulation in a different position. Norol bilim derg 2000; 17:29.
Beards, SC., Doedens, L., Jackson A., and Lipman J. (1994) A comparison of arterial lines and insertion techniques in critically ill patients. Anaesthesia, volume 49, pages 968—973
Caparas, J., Hu, J.P. and Hung, H.S. (2014) Does a novel method of PICC insertion improve safety? Nursing. 44(5), p.65-7.
Chiao, FB., Resta – Flarer, F., Lesser, J.,NG, J., Ganz, A., Pino – Luey., Bennett, H., Perkins. C. Jr., Witek, B. (2013) British Journal of Anaesthesia: Clinical Practice| Volume 110, issue 6, p966-971, June 01, 2013. Vein visualization: patient characteristic factors and efficacy of a new infrared vein finder technology.
Coomarasamy, Jeya Devi; Wint, Nyo Nyo; Saleh, Zurina Mohamad. Insertion and management of peripheral intravenous cannula in the adult medical wards of Universiti Kebangsaan Malaysia Medical Center: a best practice implementation project. JBI Database of Systematic Reviews and Implementation Reports: July 2014 – Volume 12 – Issue 7 – p 534-551 doi: 10.11124/jbisrir-2014-1672
Cooke M, Ullman AJ, Ray-Barruel G, Wallis M, Corley A, Rickard CM (2018) Not “just” an intravenous line: Consumer perspectives on peripheral intravenous cannulation (PIVC). An international cross-sectional survey of 25 countries. PLoS ONE 13(2): e0193436. https://doi.org/10.1371/journal.pone.0193436
Chen YM, Fan XW, Liu MH, Wang J, Yang YQ, Su YF. Risk factors for peripheral venous catheter failure: A prospective cohort study of 5345 patients. J Vasc Access. 2022 Nov;23(6):911-921. doi: 10.1177/11297298211015035. Epub 2021 May 13. PMID: 33985394.
DeVries, M., Valentine, M., & Mancos, P. (2016). Protected clinical indication of peripheral intravenous lines: Successful implementation. Journal of the Association for Vascular Access, 21(2), 89–92. https://doi. org/10.1016/j.java.2016.03.001
Dougherty L, Lamb J (2008) Intravenous Therapy in Nursing Practice. 2nd edn. Blackwell Publishing, Oxford
Easterlow D, Hoddinott P, Harrison S. Implementing and standardising the use of peripheral vascular access devices. J Clin Nurs. 2010 Mar;19(5-6):721-7. doi: 10.1111/j.1365-2702.2009.03098.x. PMID: 20500315.
Fabiani, A., Dreas, L., & Sanson, G. (2017). Ultrasound-guided deep-arm veins insertion of long peripheral catheters in patients with difficult venous access after cardiac surgery. Heart & Lung, 46(1), 46–53. https://doi.org/10.1016/j.hrtlng.2016.09.003
Firooz M, Karkhah S, Hosseini SJ. The effect of transilluminator device on successful peripheral venous catheter placement in children: A systematic review and meta-analysis. J Vasc Access. 2022 Oct 26:11297298221132866. doi: 10.1177/11297298221132866. Epub ahead of print. PMID: 36287005.
Hadaway L (2011) Needleless connectors: improving practice, reducing risks, Journal of the Association for Vascular Access 16(1): 20-33
Helm RE et al. (2015) Accepted but unacceptable: peripheral IV catheter failure.
Larsen, E. N., Marsh, N., O’Brien, C., Monteagle, E., Friese, C., & Rickard, C. M. (2021). Inherent and modifiable risk factors for peripheral venous catheter failure during cancer treatment: A prospective cohort study. Support Care Cancer, 29(3), 1487–1496. https://doi. org/10.1007/s00520-020-05643-
Marshburn, DM; Engelke, M. K. and Swanson M. S., “Relationships of new nurses’ perceptions and measured performance-based clinical competence,” The Journal of Continuing Education in Nursing, vol. 40, no. 9, pp. 426–432, 2009.
Marsh, N., Webster, J., Larsen, E., Cooke, M., Mihala, G., & Rickard, C. M. (2018a). Observational study of peripheral intravenous catheter outcomes in adult hospitalized patients: A multivariable analysis of peripheral intravenous catheter failure. Journal of Hospital Medicine, 13(2), 83–89. https://doi.org/10.12788/jhm.2867
Moureau, N., Sigl, G., & Hill, M. (2015). How to establish an effective midline program: A case study of 2 hospitals. Journal of the Association for Vascular Access, 20(3), 179–188. https://doi.org/10.1016/j.java. 2015.05.001
Milutinovic, D. Simin, D.and Zec, D. “Risk factor for phlebitis: ´ a questionnaire study of nurses’ perception,” Revista Latino Americana de Enfermagem, vol. 23, no. 4, pp. 677–684, 2015. Remove NFD
Meeder, A. M., Van Der Steen, A. M. Rozendaal, A. and Van Zanten, A. R. H. “Phlebitis as a consequence of peripheral intravenous administration of cisatracurium besylate in critically ill patients,” BMJ Case Reports, vol. 2016, Article ID 216448, 3 pages, 2016.
Mörgeli, R., Schmidt, K., Neumann, T. et al. A comparison of first-attempt cannulation success of peripheral venous catheter systems with and without wings and injection ports in surgical patients—a randomized trial. BMC Anesthesiol 22, 88 (2022). https://doi.org/10.1186/s12871-022-01631-7
Rickard CM, Webster J, Wallis MC Routine versus clinically indicated replacement of peripheral intravenous catheters: a randomised controlled equivalence trial. Lancet. 2012; 380:(9847)1066-1074 https://doi.org/10.1016/S0140-6736(12)61082-4
Rivera, A. M. et al. (2005) ‘The history of peripheral intravenous catheters : How little plastic tubes revolutionized medicine’, Acta Anaesthesiologica Belgica, pp. 271–282.
Schummer W, Schummer C, Gaser E, Bartunek R (2002). “Loss of the guidewire: mishap or blunder?” British journal of anaesthesia 88 (1): 144–6.
van Loon FHJ et al. (2018) Comparison of ultrasound guidance with palpation and direct visualisation for peripheral vein cannulation in adult patients: a systematic review and meta-analysis.
Zamanzadeh, V., Jasemi, M., Valizadeh, L., Keogh, B., and Taleghani, F “Lack of preparation: Iranian nurses’ experiences during transition from college to clinical practice,” Journal of Professional Nursing, vol. 31, no. 4, pp. 365–373, 2015
Marsh N, Larsen EN, O’Brien C, Ware RS, Kleidon TM, Groom P, Hewer B, Alexandrou E, Flynn J, Woollett K, Rickard CM. Safety and efficacy of midline catheters versus peripheral intravenous catheters: A pilot randomized controlled trial. Int J Nurs Pract. 2022 Oct 27:e13110. doi: 10.1111/ijn.13110. Epub ahead of print. PMID: 36303515.













Well done, Dr. Kelly. Sorry I was not more help with your writing.