Dysfunction with implantable ports is characterized by an absence of blood reflux. It can be associated to a decrease or loss of perfusion flow. Its incidence is of 0.6 to 2.5 per 1000 days/catheter.
Causes are multiple. Etiologic diagnosis is essential because each cause requires a specific treatment. They must be investigated in a precise order to avoid unnecessary medical exams.
In this article, we will discuss five implantable ports complications that can cause dysfunctions. Their identification is important as it can allow you to find suitable solutions.
-
The first dysfunctions to look for
1. Huber needle misplacement
Huber needle misplacement or secondary displacement is the first cause to eliminate. The diagnosis is clinical. When the tank of the implantable port is held between two fingers of one hand and the needle makes a lateral movement, the latter is also transmitted to the tank. If this is not the case and tank and needle are disjoint, the needle is probably misplaced.
It is important to keep in mind that this search must be careful when the port is difficult to palpate. For the most difficult cases, chest X-ray can be necessary.
2. Misplacement of the catheter tip
Misplacement of the catheter tip is the most frequent cause of blood reflux absence. It may be due to a poorly implanted central vascular access or a secondary displacement. Secondary displacements can occur with all types of vascular accesses and are favoured by intrathoracic pressure (coughing, crying for children…).
The diagnosis is based on front and side chest X-ray. The tip of the catheter must be located at the junction between the superior vena cava and the right atrium, usually 1.5 to 2.5 vertebral bodies below the carina. Profile radiography is essential to detect catheters positioned in the azygos or internal mammary vein.
In the event of malposition, the device must be changed in semi-emergency given the very high risk of thrombosis.
-
Identification of dysfunctions using dynamic imaging
If the two first causes are ruled out, the search and identification of other causes of dysfunction requires opacification of the central vascular access device with dynamic imaging. In this case, you need to look for a “fibrin sleeve”, a break in the continuity of the catheter, or a total intra-luminal occlusion of the catheter.
1. “Fibrin sleeve”
Fibrin sleeve corresponds to a mix of fibrin and fibroblast that develops at the tip of the catheter in the shape of a finger and prevents reflux. When using a chest X-ray with catheter opacification, the diameter of the device appears thicker with irregular contours. The exit of the contrast medium may appear higher than the catheter tip.
On an injected CT scan, the images can be mistaken for partial thrombus hanging from the catheter tip and thus, leading to unwarranted anticoagulation.
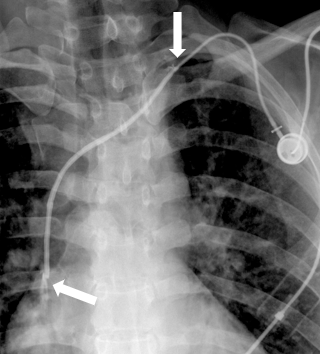 Photo 1: The two arrows are indicating the beginning and end of the fibrin sleeve
Photo 1: The two arrows are indicating the beginning and end of the fibrin sleeve
The treatment for “fibrin sleeves” is based on fibrinolytics, similar to the treatment for occlusion (that you can see below). Lasso stripping in interventional radiology is another option described in literature.
2. Break of catheter continuity
To look for catheter discontinuity, you need to look along the entire length of the catheter starting from the reservoir in the case of implantable ports. The major risk is extravasation which makes detection essential. The only possible treatment is to change the central vascular access.
3. Intra-luminal occlusion of the catheter
If the catheter’s occlusion is partial, it results in an important resistance in injection. If it is total, it leads to an impossibility of injection. In both cases, chest X-ray shows a catheter in place.
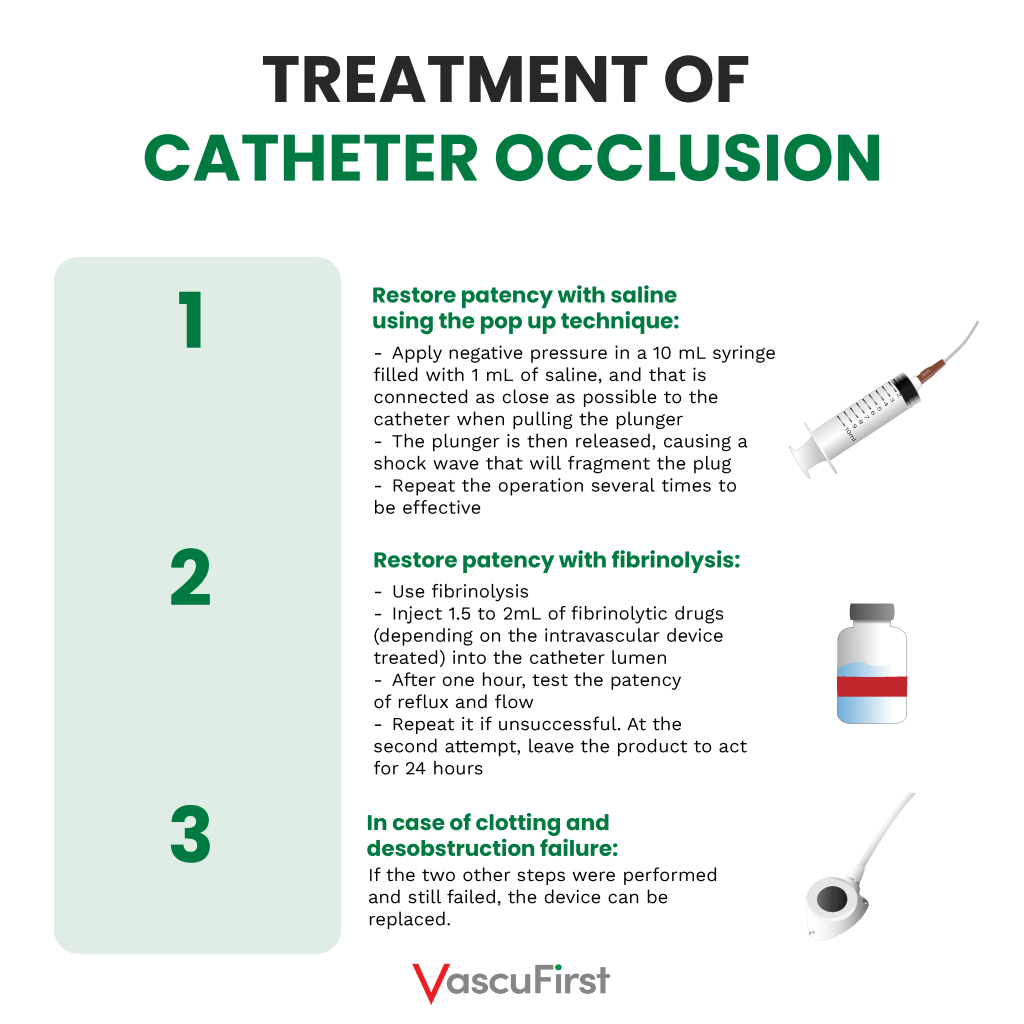
> Treatment:
> Prophylaxis:
The prevention of occlusion is fundamental. It relies on simple measures that must be protocolized and well conducted:
To conclude, dysfunction with implantable ports often manifests itself as an absence of reflux. It is important to find the aetiology of this dysfunction and to know its treatment to restore the patency when it is possible. Following the proposed diagnostic approach is also essential as it can help avoid unnecessary investigations.
Bibliography:
-
Moss et al., « Central Venous Access Devices for the Delivery of Systemic Anticancer Therapy (CAVA) ».
-
Petersen et al., « Silicone Venous Access Devices Positioned with Their Tips High in the Superior Vena Cava Are More Likely to Malfunction »
-
Gray et al., « Percutaneous Fibrin Sheath Stripping versus Transcatheter Urokinase Infusion for Malfunctioning Well-Positioned Tunneled Central Venous Dialysis Catheters »
-
Royon et al., « Experimental Study of the POP Technique »






0 Comments